GMP Compliance in Ampoule Filling Machines Explained
GMP Compliance in Ampoule Filling Machines Explained
In the pharmaceutical industry, ensuring product safety, consistency, and regulatory compliance is non-negotiable. One of the most critical frameworks governing pharmaceutical manufacturing is GMP compliance (Good Manufacturing Practices). When it comes to injectable drugs, ampoule filling machines must strictly adhere to GMP standards to guarantee sterile and high-quality production.
This comprehensive guide explains everything you need to know about GMP compliance pharma, including regulations, equipment requirements, validation processes, and best practices for achieving global compliance.
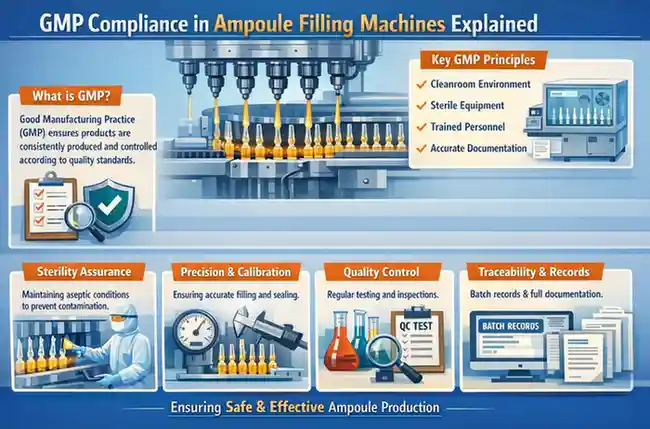
What is GMP Compliance?
Good Manufacturing Practices (GMP) are a set of regulations and guidelines that ensure pharmaceutical products are consistently produced and controlled according to quality standards.
GMP covers all aspects of production, including:
- Raw material quality
- Facility hygiene
- Equipment validation
- Staff training
- Documentation and record keeping
Why GMP Compliance is Critical for Ampoule Filling Machines
Sterile filling machines are used for injectable drugs, making compliance essential to prevent contamination and ensure patient safety.
- Prevents microbial contamination
- Ensures accurate dosing
- Maintains product integrity
- Meets regulatory approval requirements
Key GMP Requirements for Ampoule Filling Machines
1. Aseptic Design
Machines must be designed for sterile operations with minimal contamination risk.
2. Material Compliance
Use of stainless steel and non-reactive materials that meet pharmaceutical standards.
3. Cleanroom Compatibility
Equipment must operate efficiently in controlled environments.
4. Automation and Control
PLC-based systems ensure precise control and monitoring.
5. Validation and Documentation
All processes must be validated and documented for regulatory audits.
GMP Regulatory Bodies and Standards
- FDA (United States)
- EMA (Europe)
- WHO (Global guidelines)
- ISO standards
Validation Process for Ampoule Filling Machines
Validation is a key aspect of pharmaceutical regulations equipment.
Installation Qualification (IQ)
Ensures equipment is installed correctly.
Operational Qualification (OQ)
Verifies that the machine operates as intended.
Performance Qualification (PQ)
Confirms consistent performance under real production conditions.
| Validation Stage | Purpose | Outcome |
|---|---|---|
| IQ | Installation verification | Proper setup confirmed |
| OQ | Operational testing | Functionality verified |
| PQ | Performance validation | Consistent output ensured |
Quality Control Systems in GMP Compliance
Effective pharma quality control systems are essential for compliance.
- In-line inspection systems
- Leak detection technologies
- Automated rejection systems
- Batch tracking and traceability
Common Compliance Challenges
- Maintaining sterile conditions
- Meeting evolving regulatory standards
- High validation costs
- Complex documentation requirements
Cost of GMP-Compliant Ampoule Filling Machines
The cost of compliance systems is higher due to advanced features and validation requirements.
| Machine Type | Price Range (USD) | Compliance Level |
|---|---|---|
| Basic GMP Machines | $50,000 - $150,000 | Standard |
| Advanced Aseptic Systems | $150,000 - $500,000 | High |
| Fully Integrated Lines | $500,000 - $2M+ | Ultra High |
Benefits of GMP-Compliant Machines
- Regulatory approval assurance
- Improved product quality
- Reduced contamination risk
- Enhanced brand reputation
- Global market access
Integration with Smart Pharma Manufacturing
Modern compliance systems integrate with advanced technologies:
- IoT-enabled monitoring
- AI-based quality control
- Digital audit trails
- Predictive maintenance
Future Trends in GMP Compliance
- Automated compliance monitoring
- Blockchain for traceability
- AI-driven validation processes
- Smart cleanroom technologies
How to Ensure GMP Compliance
- Invest in certified equipment
- Train staff regularly
- Maintain proper documentation
- Conduct regular audits
Expert Tips
- Choose globally certified suppliers
- Focus on long-term compliance strategies
- Implement automation for consistency
ROI of GMP Compliance
Although initial investment is high, GMP compliance delivers strong returns:
| Metric | Before Compliance | After Compliance |
|---|---|---|
| Product Rejection Rate | High | Minimal |
| Regulatory Risk | High | Low |
| Market Access | Limited | Global |
Conclusion
GMP compliance in ampoule filling machines is essential for ensuring pharmaceutical quality, safety, and regulatory approval. By investing in advanced sterile filling machines and robust pharma quality control systems, manufacturers can meet global standards and build trust in their products.
As the pharmaceutical industry continues to evolve, compliance will remain a key factor in achieving long-term success and competitiveness.
Ready to achieve GMP compliance? Investing in the right equipment and processes is the foundation of a successful pharmaceutical manufacturing operation.
Comments (3)